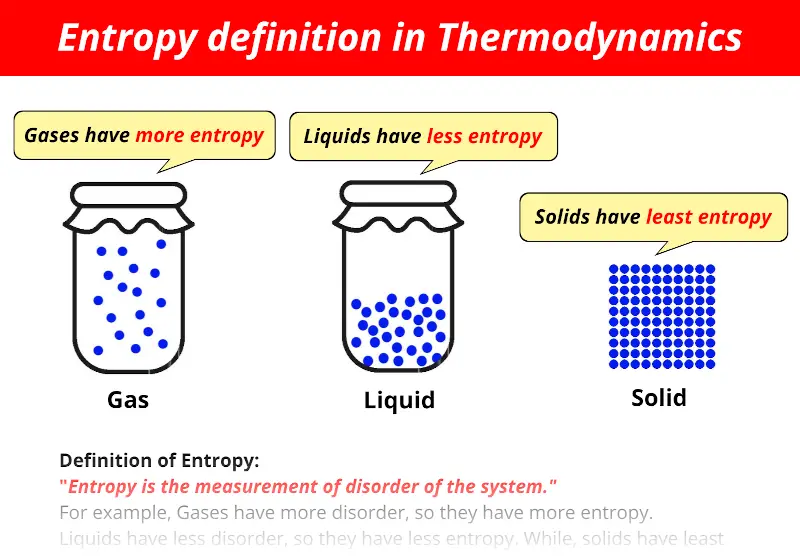
If the barriers are not very high and/or the time for the system to explore its configurational space is sufficiently large, the system will end up in a state of thermodynamic equilibrium.
#Entropy examples plus#
The system (“material plus environment”) therefore constantly samples its configurational space with a finite probability to eventually overcome barriers that separate the minima on the potential-energy surface (PES). Moreover, a material is almost always in contact with a gas (or liquid), and can exchange particles with its environment. The cluster expansion method is therefore also discussed as a numerically efficient approach for evaluating these energies.Īt finite temperatures ( T > 0 K), where functional materials typically operate, atoms move randomly in all directions due to the energy provided by heat sources.
In particular, we describe approaches for calculating the configurational density of states, which requires the evaluation of the energies of a large number of configurations. We also introduce and discuss methods for calculating phase diagrams of bulk materials and surfaces as well as point defect concentrations. We demonstrate how these concepts can be used to predict the behavior of materials at realistic temperatures and pressures within the framework of atomistic thermodynamics. In this contribution, we discuss the main concepts behind equilibrium statistical mechanics. In order to understand the properties of materials at realistic conditions, statistical effects associated with configurational sampling and particle exchange at finite temperatures must consequently be taken into account.

In most applications, functional materials operate at finite temperatures and are in contact with a reservoir of atoms or molecules (gas, liquid, or solid).
